| [1] Todo S, Demetris AJ, Van Thiel D, et al.Orthotopic liver transplantation for patients with hepatitis B virus-related liver disease. Hepatology.1991; 13(4):619-626.[2] 黄洁夫.中国肝脏移植[M].北京:人民卫生出版社,2008:676-678.[3] Takaki A, Yagi T, Iwasaki Y, et al. Short-term high-dose followed by long-term low-dose hepatitis B immunoglobulin and lamivudine therapy prevented recurrent hepatitis B after liver transplantation.Transplantation.2007;83(2):231-233.[4] 中华医学会肝病学分会,中华医学会感染病学分会.慢性乙型肝炎防治指南(2010版)[J].临床肝胆病杂志,2011,27(1):Ⅰ-ⅩⅥ.[5] 沈中阳.肝脏移植术后乙肝复发的综合防治[J].中国医学科学院报,2005,27(4):431-434.[6] Rosenaul J, MatnⅡas J,Hans L, et al.Lamivudine and low--dose hepatitis B immune globulin for prophylaxis of hepatitis B reinfection after liver transplantation possible role of mutations in the YMDD motif prior to transplantation as a risk factor for reinfection.Hepatologv.2001;34:895-902.[7] Kiyici M,Yilmaz M,Akyildiz M,et al.Association between hepatitis B and hepatocellular carcinoma recurrence in patients undergoing liver transplantation. Transplant Proc. 2008;40: 1511-1517.[8] Shouval D, Samuel D. Hepatitis B immune globulin to prevent hepatitis B virus graft reinfection following liver transplantation: a concise review. Hepatology 2000;32:1189-1195.[9] Samuel D, Muller R, Alexander G, et al. Liver transplantation in European patients with the hepatitis B surface antigen. N Engl J Med. 1993;329:1842-1847.[10] Terrault N, Roche B, Samuel D. Management of the hepatitis B virus in the liver transplantation setting: a European and an American perspective. Liver Transpl. 2005;11:716-732.[11] Omata M. Significance of extra hepatic replication of hepatitis B virus.Hepatol. 1990; 12 (2): 364-366.[12] Fang Q, Zhao LS, Zhou TY, et al.Reinfection of HBV and its possible mechanism in patients with liver transplantation. Chin J Hepatol. 2005;13 (3): 225-227.[13] Joya-Vazquez PP, Dodson FS, Dvorchik I, et al. Impact of anti-hepatitis B c-positive grafts on the outcome of liver transplantation for HBV-related cirrhosis. Transplantation. 2002;73:1598-1602.[14] Doson SF, Issa S, Araya V, et al. Infectivity of hepatitis allografts with antibodies to hepatitis B virus. Transplantation. 1997; 11:1582-1584.[15] Murray KF, Carithers RL, Jr, AASLD. Practice Guidelines: Evaluation of the Patient for Liver Transplantation. Hepatology. 2005;41(6):1407-1432.[16] Lok AS, Heathcote EJ, Hoofnagle JH.Management of hepatitis B: summary of a workshop.Gastroenterology.2001; 120(7):1828-1853.[17] Saab S, Kim M, Wright TL, et al. Successful orthotopic liver transplantation for lamivudine-associated YMDD mutant hepatitis B virus. Gastroenterology. 2000;119(5): 1382-1384.[18] Wong SN, Chu CJ, Wai CT, et al. Low risk of hepatitis B virus recurrence after withdrawal of long-term hepatitis B Immunoglobulin in patients receiving maintenance nucleoside analogue therapy. Liver Transpl. 2007; 13(3): 374-381.[19] Seehofer D. Preoperative antiviral treatment and postoperative prophyaxis in HBV DNA positive patients undergoing liver transplantation.Liver Transpl. 2001; 72:1381-1385.[20] European Association For The Study Of The Liver. EASL Clinical Practice Guidelines: management of chronic hepatitis B. J Hepatol.2009;50:227-242.[21] Liaw YF,Leung N,Kao JH,et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2008 update. Hepatol Int.2008;2:263-283.[22] 郑俊福,韩大康,李丽,等.恩替卡韦联合乙型肝炎免疫球蛋白预防肝移植术后乙型肝炎再感染[J].世界华人消化杂志,2009,17(7): 716-719.[23] 蔡常结,赵辉,郭怡,等.恩替卡韦预防肝移植术后乙型肝炎病毒再感染的临床研究[J].中华普通外科文献:电子版,2008,2(5): 385-389.[24] Samuelson A, Morgan M, Reynolds J. The use of entecavir following liver transplantation: pilot safety and tolerability data. Am J Gastroenterol. 2008;103:S160.[25] Samuel D, Forns X, Berenguer M, et al. Report of the monothematic EASL conference on liver transplantation for viral hepatitis (Paris, France, January 12-14, 2006). Hepatology. 2006;45: 127-143.[26] Chang TT, Gish RG, de Man R,et al. A comparison of entecavir and lamivudine for HBeAg-positive chronic hepatitis B. N Engl J Med.2006; 354: 1001-1010.[27] Lai CL, Rosmawati M, Lao J, et al. Entecavir is superior to lamivudine in reducing hepatitis B virus DNA in patients with chronic hepatitis B infection. Gastroenterology. 2002;123: 1831-1838.[28] Tenney DJ, Levine SM, Rose RE, et al. Clinical emergence of entecavir-resistant hepatitis B virus requires additional substitutions in virus already resistant to Lamivudine. Antimicrob Agents Chemother.2004;48:3498-3507.[29] Tenney DJ, Rose RE, Baldick CJ, et al. Long-term monitoring shows hepatitis B virus resistance to entecavir in nucleoside-naive patients is rare through 5 years of therapy. Hepatology.2009;49:1503-1514.[30] Fontana RJ, Hann HW, Wright T, et al. A multicenter study of lamivudine treatment in 33 patients with hepatitis B after liver transplantation. Liver Transpl.2001;7:504-510.[31] Mutimer D, Feraz-Neto BH, Harrison R, et al. Acute liver graft failure due to emergence of lamivudine resistant hepatitis B virus: rapid resolution during treatment with adefovir. Gut. 2001;49:860-863.[32] Lai CL, Shouval D, Lok AS, et al. Entecavir versus lamivudine for patients with HBeAg- negative chronic hepatitis B. N Engl J Med. 2006;354(10):1011- 1020.[33] Chang TT, Gish RG, de Man R, et al. A comparison of entecavir and lamivudine for HBeAg positive chronic hepatitis B. N Engl J Med. 2006;354: 1001-1010.[34] Marion P, Hann HW, Paul M, et al.Adefovir Dipivoxil (ADV) Alone And In Combination With Lamivudine (LAM) Suppresses YMDD Mutant Hepatitis B Virus Replication: 48 Week Preliminary Analysis. Abstract 845. 53rd AASLD. 2002.Boston, MA. Hepatology.2002;36(Sup 1):6-7. |
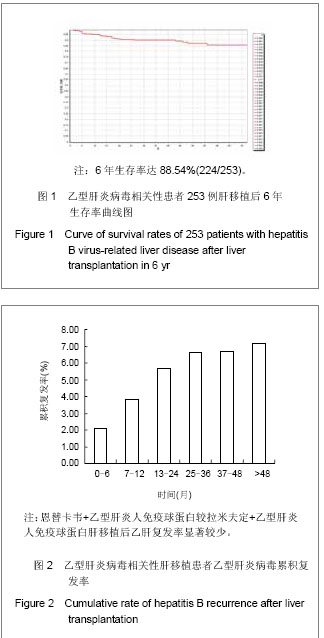
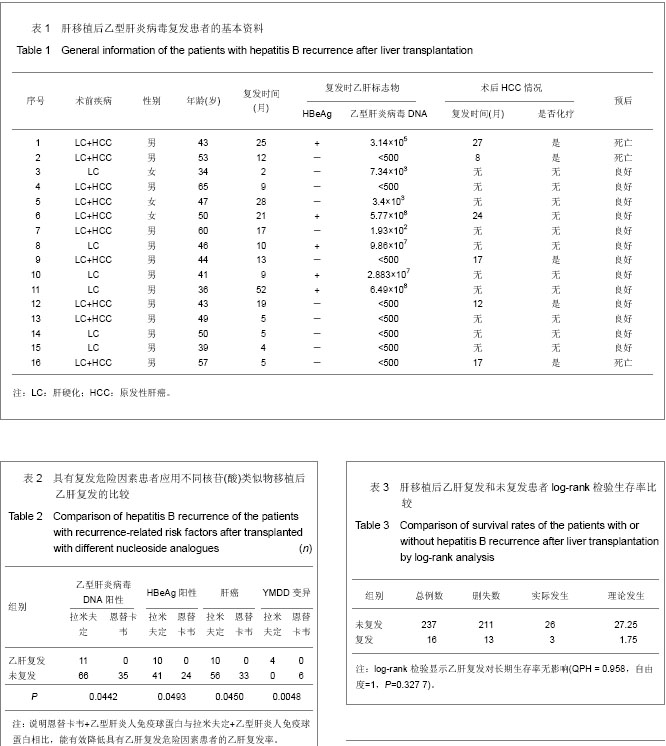
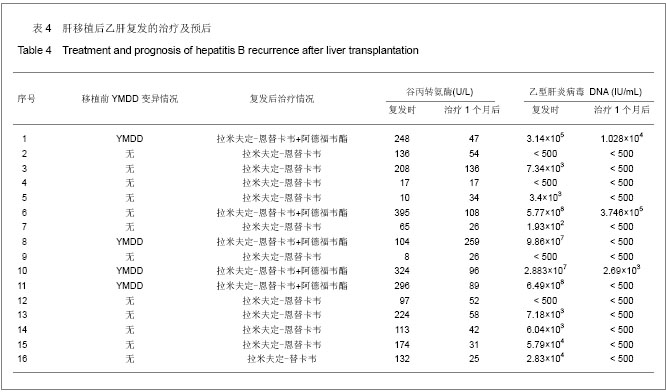
.jpg)